Salvia Chemistry
Salvinorin A (chemical formula C23H28O8) is the molecule responsible for the main psychotropic effects of the Salvia divinorum plant. This compound is present in the dried plant at about 0.18%. Active doses start as low as 100-250 micrograms sublingual; 200-500 micrograms vaporized and inhaled. This translates to 10 times the potency of psilocybin from mushrooms used in similar ways by other Mexican Indians, and more than 1000 times the potency of mescaline, from the peyote cactus (Lophophora williamsii) used as a visionary drug by the Huichol, Tarahumara and other native peoples of northern Mexico.
Unique in it’s structure, salvinorin A is considered a diterpenoid and not an alkaloid (opiates fall into this category), due to it’s lack of nitrogen atoms. Morphine, for example, contains carbon, hydrogen, oxygen, and nitrogen. Since salvinorin A only has carbon, hydrogen, and oxygen, it is cannot be broken down into salt as opiates can. It is reported to be the most potent, naturally occurring hallucinogen. Terpenoids, also called isoprenoids, are naturally occurring organic lipids that are found in all living things. Other terpenoids in Salvia divinorum have been isolated and tested, none have yet been linked to the psychoactive aspects of the plant.
There are three main opioid receptor groups called: mu, kappa, and delta. Depending on which ones are activated, different reactions can occur. Research has shown that salvinorin A is a potent κ-opioid (kappa) receptor agonist; meaning that it binds to the receptor and triggers a response by the corresponding cell. Agonists often have the same effect as a naturally occurring substance. The κ-opioid receptor is a protein and one of five receptors that bind opium-like compounds in the brain, controlling the effects of said compounds(including mood, consciousness, motor control, and pain). It also is the main receptor associated with visual hallucinations. Opiates such as morphine also weakly trigger the k-opioid receptor, but also strongly activate the mu-opioid receptor (the receptor which is thought to produce the addictive quality). Salvinorin A triggers only the k-opioid receptor, inducing strong visual hallucinations, without the addictive quality common to other opiates.
Salvinorin A has a high affinity for the receptor, indicated by the low dissociation constant of 1.0 nanomolar (nM), meaning it makes a strong paired bond. It has been reported that the effects of salvinorin A in mice are blocked by κ-opioid receptor antagonists. This makes it unlikely that another mechanism contributes independently to the compound’s observed effects in mice. Salvinorin A has also recently been found to act as a dopamine D2 receptor partial agonist, with an affinity of 5–10 nM, an intrinsic activity of 40–60%, and an EC50 of 50–90 nM, which is several-fold higher than its EC50 of 235 nM for the κ-opioid receptor. The EC50 is a measurement derived from when the concentration of a substance’s response is halfway between the baseline and maximum after some specified exposure time, i.e. 50% of max response. This suggests that the D2 receptor may also play an important role in its effects.
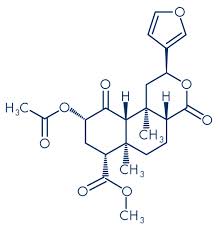
Molecular structure of salvinorum A
Molecular formula: C23H28O8
Molecular weight: 432.47 g/mol
Dopamine receptors range from 5 confirmed (D1 to D5) along with a possibility of at least 2 more (D6-D7). They are all ubiquitous throughout the brain, D1-D2 receptors being found in quantities 10-100 times that of D3-D5. Dopamine, being the main neurotransmitter concerning reward pathways in the brain, has long been associated with euphoric effects and drug use. Many drugs (recreational and prescribed) change the way dopamine is processed, usually limiting the ability of the DAT (dopamine transporter protein) to flush out the dopamine, in turn causing an increase of both dopamine and related signaling (mostly in the D1 and D2 receptors).
There isn’t much data about what dosages cause damage, because even at high doses salvia is non-toxic. Pulse pressure has been reported to elevate slightly (but remained statistically insignificant about 1.5-2 times the control group) and mice chronically given an extremely large dosage did not show signs of organ damage. People who smoked 580 micrograms of the pure drug had urine salvinorin A concentrations of 2.4–10.9 micrograms per liter during the first hour, but the levels fell below the detection limit by 1.5 hours after smoking, indicating that it moves out of the system quickly, or at least at any currently detectable levels.